what happens to gibbs energy with pressure increase
In thermodynamics, the Gibbs energy (or Gibbs free energy) is a thermodynamic potential that tin can be used to summate the maximum reversible work that may exist performed by a thermodynamic arrangement at a constant temperature and force per unit area. The Gibbs gratis free energy ( , measured in joules in SI) is the maximum amount of non-expansion work that can be extracted from a thermodynamically airtight arrangement (one that can exchange heat and work with its surroundings, simply not matter). This maximum can be attained only in a completely reversible process. When a organisation transforms reversibly from an initial country to a concluding country, the decrease in Gibbs free energy equals the work done by the system to its surroundings, minus the work of the pressure forces.[i]
The Gibbs energy (symbol ) is besides the thermodynamic potential that is minimized when a organisation reaches chemical equilibrium at constant pressure and temperature. Its derivative with respect to the reaction coordinate of the organisation vanishes at the equilibrium point. Equally such, a reduction in is necessary for a reaction to be spontaneous at constant force per unit area and temperature.
The Gibbs free energy, originally called available energy, was developed in the 1870s by the American scientist Josiah Willard Gibbs. In 1873, Gibbs described this "bachelor free energy" as[two] : 400
the greatest corporeality of mechanical work which tin be obtained from a given quantity of a certain substance in a given initial country, without increasing its total book or allowing heat to laissez passer to or from external bodies, except such as at the close of the processes are left in their initial status.
The initial country of the body, according to Gibbs, is supposed to be such that "the body can be made to pass from information technology to states of dissipated energy by reversible processes". In his 1876 magnum opus On the Equilibrium of Heterogeneous Substances, a graphical analysis of multi-phase chemical systems, he engaged his thoughts on chemical-free free energy in full.
If the reactants and products are all in their thermodynamic standard states, then the defining equation is written equally .
Overview [edit]

The reaction C(s) diamond → C(s) graphite has a negative alter in Gibbs gratis free energy and is therefore thermodynamically favorable at 25 °C and 1 atm. Still, the reaction is too tiresome to be observed, because of its very high activation energy. Whether a reaction is thermodynamically favorable does not determine its rate.
Co-ordinate to the 2nd law of thermodynamics, for systems reacting at standard weather for temperature and pressure (or any other fixed temperature and force per unit area), at that place is a general natural trend to achieve a minimum of the Gibbs free energy.
A quantitative mensurate of the favorability of a given reaction at constant temperature and pressure is the change ΔM (sometimes written "delta One thousand" or "dThou") in Gibbs free energy that is (or would be) caused by the reaction. Every bit a necessary condition for the reaction to occur at constant temperature and force per unit area, ΔG must be smaller than the non-force per unit area-volume (non-PV, e.g. electric) piece of work, which is often equal to nada (hence ΔG must be negative). ΔG equals the maximum amount of not-PV work that can exist performed as a result of the chemical reaction for the instance of reversible procedure. If analysis indicates a positive ΔThousand for a reaction, then energy — in the course of electrical or other not-PV work — would have to be added to the reacting organisation for ΔThou to be smaller than the non-PV work and make it possible for the reaction to occur.[3] : 298–299
One can call up of ∆Thou as the amount of "free" or "useful" energy bachelor to practice work. The equation tin be also seen from the perspective of the arrangement taken together with its environment (the rest of the universe). Get-go, one assumes that the given reaction at constant temperature and pressure is the only ane that is occurring. So the entropy released or captivated by the system equals the entropy that the environment must absorb or release, respectively. The reaction will only be allowed if the total entropy change of the universe is zero or positive. This is reflected in a negative ΔG, and the reaction is called an exergonic process.
If two chemical reactions are coupled, then an otherwise endergonic reaction (one with positive ΔG) can exist made to happen. The input of heat into an inherently endergonic reaction, such as the elimination of cyclohexanol to cyclohexene, can exist seen as coupling an unfavourable reaction (elimination) to a favourable one (burning of coal or other provision of rut) such that the full entropy change of the universe is greater than or equal to zero, making the total Gibbs free energy deviation of the coupled reactions negative.
In traditional utilize, the term "free" was included in "Gibbs energy" to mean "bachelor in the class of useful work".[ane] The characterization becomes more than precise if we add together the qualification that it is the energy available for non-pressure-volume piece of work.[four] (An analogous, but slightly different, meaning of "costless" applies in conjunction with the Helmholtz free energy, for systems at constant temperature). However, an increasing number of books and journal articles do not include the attachment "free", referring to M as merely "Gibbs energy". This is the outcome of a 1988 IUPAC coming together to prepare unified terminologies for the international scientific community, in which the removal of the adjective "free" was recommended.[v] [6] [seven] This standard, however, has not yet been universally adopted.
The name "costless enthalpy" has also been used for G in the past.[vi]
History [edit]
The quantity chosen "free energy" is a more advanced and accurate replacement for the outdated term affinity, which was used by chemists in the earlier years of physical chemical science to describe the force that caused chemical reactions.
In 1873, Josiah Willard Gibbs published A Method of Geometrical Representation of the Thermodynamic Properties of Substances past Means of Surfaces, in which he sketched the principles of his new equation that was able to predict or estimate the tendencies of various natural processes to ensue when bodies or systems are brought into contact. By studying the interactions of homogeneous substances in contact, i.e., bodies composed of role solid, part liquid, and office vapor, and past using a iii-dimensional volume-entropy-internal energy graph, Gibbs was able to make up one's mind three states of equilibrium, i.e., "necessarily stable", "neutral", and "unstable", and whether or not changes would ensue. Farther, Gibbs stated:[2]
If we wish to express in a single equation the necessary and sufficient condition of thermodynamic equilibrium for a substance when surrounded by a medium of constant pressure p and temperature T, this equation may exist written:
δ(ε − Tη + pν) = 0
when δ refers to the variation produced past any variations in the country of the parts of the torso, and (when different parts of the body are in different states) in the proportion in which the body is divided betwixt the different states. The condition of stable equilibrium is that the value of the expression in the parenthesis shall be a minimum.
In this description, equally used by Gibbs, ε refers to the internal energy of the body, η refers to the entropy of the body, and ν is the volume of the body...
Thereafter, in 1882, the German language scientist Hermann von Helmholtz characterized the affinity as the largest quantity of work which tin can be gained when the reaction is carried out in a reversible mode, e.g., electrical work in a reversible cell. The maximum work is thus regarded every bit the diminution of the free, or available, energy of the system (Gibbs gratis energy M at T = constant, P = abiding or Helmholtz gratuitous energy F at T = abiding, Five = constant), whilst the heat given out is usually a measure of the diminution of the full energy of the arrangement (internal energy). Thus, M or F is the amount of energy "complimentary" for work nether the given weather condition.
Until this signal, the full general view had been such that: "all chemic reactions drive the organisation to a state of equilibrium in which the affinities of the reactions vanish". Over the next 60 years, the term analogousness came to be replaced with the term gratis energy. Co-ordinate to chemistry historian Henry Leicester, the influential 1923 textbook Thermodynamics and the Gratis Energy of Chemic Substances by Gilbert North. Lewis and Merle Randall led to the replacement of the term "affinity" past the term "costless energy" in much of the English-speaking world.[viii] : 206
Definitions [edit]
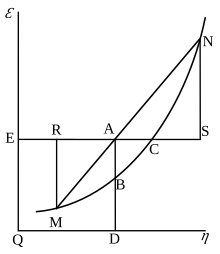
Willard Gibbs' 1873 available energy (gratuitous energy) graph, which shows a airplane perpendicular to the centrality of v (volume) and passing through point A, which represents the initial state of the torso. MN is the section of the surface of dissipated energy. Qε and Qη are sections of the planes η = 0 and ε = 0, and therefore parallel to the axes of ε (internal energy) and η (entropy), respectively. AD and AE are the energy and entropy of the body in its initial state, AB and AC its bachelor energy (Gibbs costless free energy) and its chapters for entropy (the amount by which the entropy of the body can be increased without changing the energy of the body or increasing its volume) respectively.
The Gibbs free energy is defined equally
which is the aforementioned every bit
where:
- U is the internal energy (SI unit: joule),
- p is pressure (SI unit: pascal),
- V is volume (SI unit: mthree),
- T is the temperature (SI unit: kelvin),
- S is the entropy (SI unit: joule per kelvin),
- H is the enthalpy (SI unit of measurement: joule).
The expression for the minute reversible alter in the Gibbs free energy as a function of its "natural variables" p and T, for an open system, subjected to the performance of external forces (for instance, electrical or magnetic) Teni , which cause the external parameters of the organization ai to change by an amount dai , tin be derived every bit follows from the first police for reversible processes:
where:
- μ i is the chemical potential of the i-th chemic component. (SI unit: joules per particle[9] or joules per mole[1])
- N i is the number of particles (or number of moles) composing the i-thursday chemical component.
This is one grade of Gibbs primal equation.[10] In the infinitesimal expression, the term involving the chemical potential accounts for changes in Gibbs free energy resulting from an influx or outflux of particles. In other words, it holds for an open system or for a airtight, chemically reacting system where the Due northi are changing. For a closed, not-reacting system, this term may exist dropped.
Any number of extra terms may be added, depending on the particular system being considered. Bated from mechanical work, a system may, in addition, perform numerous other types of work. For example, in the infinitesimal expression, the contractile work energy associated with a thermodynamic system that is a contractile cobweb that shortens by an corporeality −dl under a strength f would result in a term f dl beingness added. If a quantity of charge −de is caused by a organization at an electrical potential Ψ, the electrical work associated with this is −Ψ deast, which would be included in the infinitesimal expression. Other work terms are added on per arrangement requirements.[xi]
Each quantity in the equations above can be divided by the corporeality of substance, measured in moles, to form molar Gibbs free free energy. The Gibbs gratis energy is i of the almost important thermodynamic functions for the characterization of a system. Information technology is a cistron in determining outcomes such as the voltage of an electrochemical cell, and the equilibrium constant for a reversible reaction. In isothermal, isobaric systems, Gibbs free free energy can exist thought of as a "dynamic" quantity, in that information technology is a representative measure of the competing effects of the enthalpic[ clarification needed ] and entropic driving forces involved in a thermodynamic process.
Relation to other relevant parameters
The temperature dependence of the Gibbs energy for an platonic gas is given by the Gibbs–Helmholtz equation, and its pressure dependence is given past[12]
or more than conveniently equally its chemic potential:
In non-ideal systems, fugacity comes into play.
Derivation [edit]
The Gibbs gratuitous energy total differential with respect to natural variables may be derived past Legendre transforms of the internal energy.
The definition of M from higher up is
- .
Taking the total differential, we have
Replacing dU with the issue from the first police gives[xiii]
The natural variables of G are then p, T, and {N i }.
Homogeneous systems [edit]
Because S, 5, and Due north i are all-encompassing variables, an Euler integral allows easy integration of dU:[13]
Because some of the natural variables of Thousand are intensive, dG may not be integrated using Euler integrals as is the case with internal free energy. All the same, simply substituting the above integrated outcome for U into the definition of G gives a standard expression for G:[13]
This result applies to homogeneous, macroscopic systems, but not to all thermodynamic systems.[14]
Gibbs free free energy of reactions [edit]
The system under consideration is held at constant temperature and pressure, and is closed (no affair can come up in or out). The Gibbs energy of whatsoever arrangement is and an infinitesimal modify in One thousand, at constant temperature and pressure yields:
By the first law of thermodynamics, a change in the internal energy U is given by
where δQ is energy added as heat, and δW is energy added every bit work. The work done on the system may be written every bit δW = −PdV + δW10 , where −PdV is the mechanical work of pinch/expansion washed on the system and δW10 is all other forms of work, which may include electrical, magnetic, etc. Assuming that simply mechanical work is done,
and the infinitesimal alter in G is:
The 2nd law of thermodynamics states that for a closed arrangement, , and and so it follows that:
This means that for a system which is not in equilibrium, its Gibbs free energy will ever exist decreasing, and when it is in equilibrium (i.east. no longer changing), the infinitesimal alter dG will be nil. In particular, this will be true if the system is experiencing whatsoever number of internal chemical reactions on its path to equilibrium.
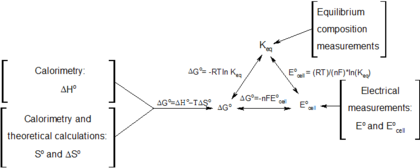
In electrochemical thermodynamics [edit]
When electric accuse dQ is passed in an electrochemical prison cell the emf ℰ yields a thermodynamic work term that appears in the expression for the change in Gibbs energy:
where G is the Gibbs' free energy, S is the entropy, 5 is the system book, P is its pressure level and T is its absolute temperature.
The combination (ℰ, Q) is an example of a conjugate pair of variables. At constant pressure the above relationship produces a Maxwell relation that links the change in open up cell voltage with temperature T (a measurable quantity) to the change in entropy S when charge is passed isothermally and isobarically. The latter is closely related to the reaction entropy of the electrochemical reaction that lends the bombardment its ability. This Maxwell relation is:[15] [ commendation needed ]
If a mole of ions goes into solution (for example, in a Daniell cell, equally discussed below) the accuse through the external circuit is:
where north 0 is the number of electrons/ion, and F 0 is the Faraday abiding and the minus sign indicates belch of the cell. Bold constant pressure and book, the thermodynamic backdrop of the jail cell are related strictly to the beliefs of its emf by:
where ΔH is the enthalpy of reaction. The quantities on the correct are all directly measurable.
Useful identities to derive the Nernst equation [edit]
During a reversible electrochemical reaction at abiding temperature and pressure, the following equations involving the Gibbs gratuitous free energy hold:
and rearranging gives
which relates the cell potential resulting from the reaction to the equilibrium constant and reaction quotient for that reaction (Nernst equation),
where
- Δr G , Gibbs free energy alter per mole of reaction,
- Δr One thousand° , Gibbs free energy change per mole of reaction for unmixed reactants and products at standard weather condition (i.e. 298K, 100kPa, 1M of each reactant and product),
- R , gas abiding,
- T , absolute temperature,
- ln, natural logarithm,
- Q r , reaction quotient (unitless),
- K eq , equilibrium abiding (unitless),
- due west elec,rev , electric work in a reversible process (chemistry sign convention),
- n , moles of electrons transferred in the reaction,
- F = N A q e ≈ 96485C/mol, Faraday abiding (charge per mole of electrons),
- E , prison cell potential,
- E° , standard cell potential.
Moreover, we likewise have:
which relates the equilibrium constant with Gibbs free free energy. This implies that at equilibrium
and
Standard energy change of germination [edit]
| Substance (state) | Δ f G° | |
|---|---|---|
| (kJ/mol) | (kcal/mol) | |
| NO(g) | 87.six | 20.ix |
| NO2(thou) | 51.3 | 12.3 |
| NorthiiO(g) | 103.vii | 24.78 |
| H2O(g) | −228.6 | −54.64 |
| H2O(l) | −237.1 | −56.67 |
| CO2(k) | −394.4 | −94.26 |
| CO(g) | −137.2 | −32.79 |
| CH4(g) | −50.5 | −12.ane |
| C2Hvi(chiliad) | −32.0 | −7.65 |
| C3H8(g) | −23.iv | −v.59 |
| CviH6(g) | 129.7 | 29.76 |
| C6Hhalf-dozen(l) | 124.5 | 31.00 |
The standard Gibbs energy of formation of a compound is the change of Gibbs free energy that accompanies the formation of 1 mole of that substance from its component elements, at their standard states (the most stable course of the element at 25 °C and 100 kPa). Its symbol is Δ f G˚.
All elements in their standard states (diatomic oxygen gas, graphite, etc.) have standard Gibbs free energy modify of formation equal to zero, as there is no change involved.
- Δf K = Δ f Yard˚ + RT ln Qf ,
where Qf is the reaction quotient.
At equilibrium, Δ f G = 0, and Qf = Thou, so the equation becomes
- Δ f G˚ = −RT ln K,
where K is the equilibrium abiding.
Graphical interpretation by Gibbs [edit]
Gibbs free free energy was originally defined graphically. In 1873, American scientist Willard Gibbs published his first thermodynamics paper, "Graphical Methods in the Thermodynamics of Fluids", in which Gibbs used the two coordinates of the entropy and volume to represent the state of the body. In his 2d follow-up paper, "A Method of Geometrical Representation of the Thermodynamic Properties of Substances by Means of Surfaces", published later that year, Gibbs added in the third coordinate of the energy of the body, defined on three figures. In 1874, Scottish physicist James Clerk Maxwell used Gibbs' figures to brand a 3D free energy-entropy-volume thermodynamic surface of a fictitious h2o-like substance.[17] Thus, in order to understand the concept of Gibbs free energy, it may aid to empathise its interpretation by Gibbs as section AB on his figure three, and as Maxwell sculpted that section on his 3D surface figure.
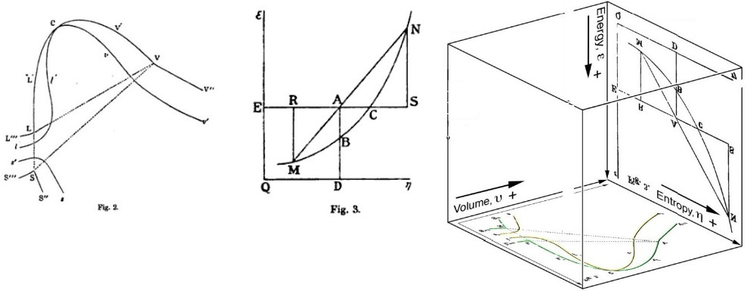
American scientist Willard Gibbs' 1873 figures ii and three (above left and eye) used past Scottish physicist James Clerk Maxwell in 1874 to create a 3-dimensional entropy, volume, energy thermodynamic surface diagram for a fictitious h2o-like substance, transposed the two figures of Gibbs (above right) onto the volume-entropy coordinates (transposed to bottom of cube) and energy-entropy coordinates (flipped upside down and transposed to back of cube), respectively, of a three-dimensional Cartesian coordinates; the region AB being the first-ever 3-dimensional representation of Gibbs free energy, or what Gibbs chosen "bachelor energy"; the region AC being its capacity for entropy, what Gibbs defined as "the amount by which the entropy of the body can be increased without changing the energy of the torso or increasing its book.
See also [edit]
- Bioenergetics
- Calphad (Adding of Phase Diagrams)
- Critical point (thermodynamics)
- Electron equivalent
- Enthalpy-entropy compensation
- Free entropy
- Gibbs–Helmholtz equation
- Grand potential
- Non-random two-liquid model (NRTL model) – Gibbs energy of backlog and mixing calculation and action coeffients
- Spinodal – Spinodal Curves (Hessian matrix)
- Standard molar entropy
- Thermodynamic energy
- UNIQUAC model – Gibbs energy of backlog and mixing calculation and activity coeffients
Notes and references [edit]
- ^ a b c Perrot, Pierre (1998). A to Z of Thermodynamics. Oxford University Printing. ISBN0-19-856552-6.
- ^ a b Gibbs, Josiah Willard (December 1873). "A Method of Geometrical Representation of the Thermodynamic Properties of Substances by Means of Surfaces" (PDF). Transactions of the Connecticut Academy of Arts and Sciences. two: 382–404.
- ^ Peter Atkins; Loretta Jones (1 August 2007). Chemical Principles: The Quest for Insight . West. H. Freeman. ISBN978-ane-4292-0965-6.
- ^ Reiss, Howard (1965). Methods of Thermodynamics. Dover Publications. ISBN0-486-69445-3.
- ^ Calvert, J. Grand. (1 Jan 1990). "Glossary of atmospheric chemical science terms (Recommendations 1990)". Pure and Applied Chemistry. 62 (11): 2167–2219. doi:x.1351/pac199062112167.
- ^ a b "Gibbs energy (function), Chiliad". IUPAC Gold Book (Compendium of Chemical Technology). IUPAC (International Union of Pure and Practical Chemical science). 2008. doi:x.1351/goldbook.G02629. Retrieved 24 Dec 2020.
Information technology was formerly called free free energy or free enthalpy.
- ^ Lehmann, H. P.; Fuentes-Arderiu, X.; Bertello, 50. F. (1 Jan 1996). "Glossary of terms in quantities and units in Clinical Chemistry (IUPAC-IFCC Recommendations 1996)". Pure and Applied Chemical science. 68 (four): 957–1000. doi:10.1351/pac199668040957. S2CID 95196393.
- ^ Henry Marshall Leicester (1971). The Historical Groundwork of Chemistry. Courier Corporation. ISBN978-0-486-61053-five.
- ^ Chemic Potential, IUPAC Gold Book.
- ^ Müller, Ingo (2007). A History of Thermodynamics – the Doctrine of Free energy and Entropy. Springer. ISBN978-3-540-46226-2.
- ^ Katchalsky, A.; Curran, Peter F. (1965). Nonequilibrium Thermodynamics in Biophysics. Harvard University Printing. CCN 65-22045.
- ^ Atkins, Peter; de Paula, Julio (2006). Atkins' Concrete Chemistry (8th ed.). W. H. Freeman. p. 109. ISBN0-7167-8759-viii.
- ^ a b c Salzman, William R. (2001-08-21). "Open Systems". Chemical Thermodynamics. Academy of Arizona. Archived from the original on 2007-07-07. Retrieved 2007-x-xi .
- ^ Brachman, M. K. (1954). "Fermi Level, Chemical Potential, and Gibbs Free Energy". The Journal of Chemical Physics. 22 (6): 1152. Bibcode:1954JChPh..22.1152B. doi:10.1063/ane.1740312.
- ^ H. South. Harned, B. B. Owen, The Physical Chemical science of Electrolytic Solutions, third edition, Reinhold Publishing Corporation, North.Y.,1958, p. ii-6
- ^ CRC Handbook of Chemical science and Physics, 2009, pp. v-4–five-42, 90th ed., Lide.
- ^ James Clerk Maxwell, Elizabeth Garber, Stephen Yard. Brush, and C. W. Francis Everitt (1995), Maxwell on heat and statistical mechanics: on "fugitive all personal enquiries" of molecules, Lehigh Academy Press, ISBN 0-934223-34-3, p. 248.
External links [edit]
- IUPAC definition (Gibbs free energy)
- Gibbs free free energy calculator
- Gibbs free energy – Florida State University
- Gibbs Costless Energy – Eric Weissteins World of Physics
- Entropy and Gibbs Free Energy – www.2ndlaw.oxy.edu
- Gibbs Costless Energy – Georgia State University
- Gibbs Gratis Energy Java Applet – University of California, Berkeley
- Using Gibbs Energy for prediction of chemical driven material ageing
- Topological Analysis of the Gibbs Energy Function (Liquid-Liquid Equilibrium Correlation Data). Including a Thermodinamic Review and a Graphical User Interface (GUI) for Surfaces/Tie-lines/Hessian matrix analysis – University of Alicante (Reyes-Labarta et al. 2015–xviii)
Source: https://en.wikipedia.org/wiki/Gibbs_free_energy





























0 Response to "what happens to gibbs energy with pressure increase"
Post a Comment